
Transcranial Temporal Interference Stimulation
Temporal interference (TI) Stimulation is a non-invasive brain stimulation method designed to reach deeper brain regions without surgery. It works by applying two high-frequency electrical currents (in the range of kilohertz) through electrodes placed on the scalp. These currents pass harmlessly through the outer brain but interact with each other deep inside, creating a low-frequency “beat” pattern where they overlap. This low-frequency signal can modulate the activity of specific brain circuits while minimizing stimulation of tissues closer to the scalp. TI is being investigated as a way to more precisely and comfortably target networks involved in mood, cognition, and other mental health conditions.
Current Studies
Project 3: Temporal Interference Stimulation for Major Depressive Disorder: A Pilot Randomized Control Trial: Recruiting
Overview:
We are conducting a clinical trial testing temporal interference (TI) stimulation to help people living with major depressive disorder (MDD). This study focuses on a brain region called the subgenual anterior cingulate cortex (sgACC), which plays an important role in mood and emotional regulation. Participants will receive non-invasive TI stimulation while completing simple mood-related activities, such as watching short emotional video clips, across 10 sessions. By studying how this stimulation affects brain activity and mood, we hope to develop new, more targeted treatment options for depression in the future.
Our lab is currently recruiting participants for this important study. If you’re interested in contributing to this innovative research, please contact TIStimulation@unityhealth.to or call the study coordinator Ilya at 416-360-4000 (ext. 77062).
Eligibility:
Inclusion Criteria
- Any sex or gender, aged 18 to 65 years
- Diagnosis of Major Depressive Disorder
- Able to speak and understand English
Exclusion Criteria
- History of bipolar disorder or psychosis
- Current substance use or history of substance use during the past year (excluding nicotine and caffeine use disorder)
- Certain medical illnesses and blood test results
- Recent change in treatment regimen for depression than happened less than 1 month ago
Treatment description:
- You will receive 10 sessions of TI stimulation or placebo over 2 weeks (weekdays only). Each session lasts 30–45 minutes and involves watching film clips during the session.
- On select days, we will record brain activity using electroencephalography (EEG), a safe and non-invasive procedure.
- You will complete two MRI scans — one before you start and one after you finish 10 days of treatment — each lasting about 1 hour.
- We will follow up with you for 1 month after completing treatment to monitor longer-term effects.
Functional Electrical Stimulation of Facial Muscles
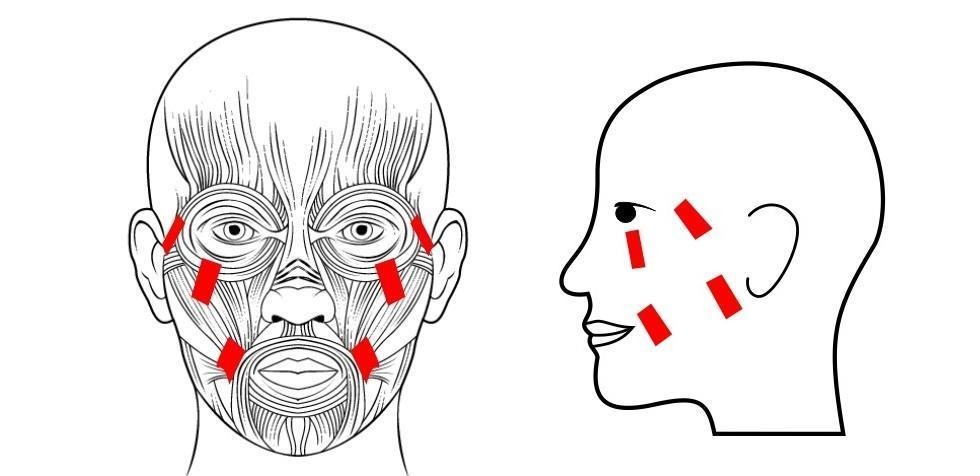
Project 1: “Take-Home” Functional Electrical Stimulation (FES) for Depression: Recruiting
Overview:
Bilateral functional electrical stimulation (FES) of the facial muscles involves stimulating specific facial muscles with an FES device and is being investigated as a potential novel intervention for major depressive disorder (MDD). Applying FES on specific facial muscles related to smile patterns may result in better control of emotions and mood elevation. FES is a low-risk, non-invasive technique which causes muscles to contract using electrical current. The purpose of this study is to learn whether FES of the facial muscles is effective in treating MDD. This study is a home-based study and all the treatment visits will be done by the participants at home. For this purpose we will make a personalized mask for each participant.
Our lab is currently recruiting participants for this important study. If you’re interested in contributing to this innovative research, please contact the study team at Take-Home-FES@unityhealth.to or call 416-360-4000 (ext. 40675).
Eligibility:
Inclusion Criteria:
- Diagnosis of Major Depressive Disorder
- Male or non pregnant female, above 18 years of age
- No change in the medication regimen or other forms of treatments for at least 4 weeks prior to beginning the study, during the 20-session treatment period, and the 4-week post-treatment observation period.
Exclusion Criteria:
- Have a history of epilepsy or seizures
- Have any damage or dysfunction of facial nerves
Treatment description:
In this study, we will be applying FES to specific facial muscles. This electrical current may cause gradual, long-lasting changes in brain activity levels due to the physiological connection existing between facial muscles and the brain.
- Personalized masks will be manufactured and delivered to participants.
- Participants will be randomized to receive either 20 home-based sessions (45 minutes) of FES or sham stimulation over 4 weeks.
- Depressive symptoms will be assessed pre-treatment, after every five treatment days, and during the post-stimulation visits.
Project 2: Functional Electrical Stimulation for Major Depressive Disorder: A Pilot Randomized Control Trial: Recruiting
Overview:
Bilateral functional electrical stimulation (FES) of the facial muscles involves stimulating specific facial muscles with an FES device and is being investigated as a potential novel intervention for major depressive disorder (MDD). Applying FES on specific facial muscles related to smile patterns may result in better control of emotions and mood elevation. FES is a low-risk, non-invasive technique which causes muscles to contract using electrical current. The purpose of this study is to learn whether FES of the facial muscles is effective in treating MDD. This study aims to assess the effectiveness, tolerability, and safety of FES in patients with MDD.
Our lab is currently recruiting participants for this important study. If you’re interested in contributing to this innovative research, please contact the study team at Take-Home-FES@unityhealth.to or call 416-360-4000 (ext. 40675).
Eligibility:
Inclusion Criteria:
- Diagnosis of Major Depressive Disorder
- Male or non pregnant female, aged 18 to 70 years old
- No change in the medication regimen or other forms of treatments for at least 4 weeks prior to beginning the study, during the 20-session treatment period, and the 4-week post-treatment observation period.
Exclusion Criteria:
- Have a history of epilepsy or seizures
- Have any damage or dysfunction of facial nerves
Treatment description:
In this study, we will be applying FES to specific facial muscles. This electrical current may cause gradual, long-lasting changes in brain activity levels due to the physiological connection existing between facial muscles and the brain.
- Participants will be randomized to receive either 20 sessions (45 minutes- twice per day) of FES or sham stimulation over 2 weeks.
- Depressive symptoms will be assessed pre-treatment, at the end of each treatment week, and during the post-stimulation visits.
- Here is the study design:
